Digital infrared thermal imaging of body and hoof skin surface temperature profile in Murrah buffaloes (Bubalus bubalis): A preliminary report
DOI:
https://doi.org/10.56825/bufbu.2022.4143463Keywords:
Bubalus bubalis, buffaloes, Infrared thermography, Murrah buffaloes, ambient temperature, hoof, temperature, hoof healthAbstract
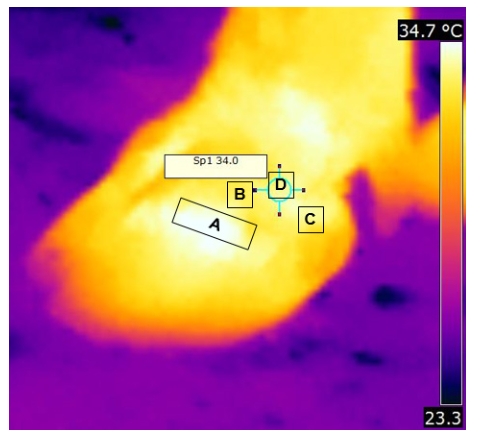
The aim of the present study was to generate thermographic profile of foot region viz. body temperature (BTºC), coronary band temperature (CBTºC), hoof skin surface temperature (HSSTºC) and mean temperature difference (ΔTºC) in Murrah buffaloes (Bubalus bubalis) and its relation with ambient temperature. A total of 60 hooves of lactating Murrah buffaloes (n = 15) maintained in a farm, were monitored once a day continuously for 15 days before evening milking using forward-looking infrared (FLIR) i5 camera. Total of 1125 thermograms were analyzed using FLIR Quick Report 1.2 image analysis software. The mean ± SD (°C) of BT, CBT, HSST and ΔT of all animals throughout the experimental period were 37.39±0.05, 34.93±0.13, 34.16±0.13 and 0.76±0.26 respectively. The CBT was around 0.5 to 1°C higher than HSST. Statistical analysis revealed that ambient temperature is highly and positively correlated with CBT (ºC) (r = 0.95) and HSST (ºC) (r = 0.99), indicating greater influence of ambient temperature on surface temperature of foot region. Therefore, CBT (ºC) and HSST (ºC) have a tendency to follow variations in ambient temperature (R2 = 0.91 and 0.98 respectively). However, significant difference was observed between BT vs. CBT, BT vs. HSST and CBT vs. HSST (ºC). The mean ± SD (°C) of ΔT of all animals, throughout the observation period was 0.96±0.41. A significant difference was observed in ΔT between animals (P<0.001). However, significant difference was not observed in ΔT between days and among four hooves within animal. It is concluded that, to the best of our knowledge this is the first report on baseline thermographic information on BT, CBT, HSST and ΔT differentials for buffalo and its clinical significance in monitoring hoof health and management.
Downloads
Metrics
References
Ahirwar, M.K., M.A. Kataktalware, H.A. Pushpadass, S. Jeyakumar, S. Jash, S. Nazar, G.L. Devi, J.P. Kastelic and K.P. Ramesha. 2018. Scrotal infrared digital thermography predicts effects of thermal stress on buffalo (Bubalus bubalis) semen. J. Therm. Biol., 78: 51-57. DOI: 10.1016/j.jtherbio.2018.09.003
Alsaaod, M. and W. Buscher. 2012. Detection of hoof lesions using digital infrared thermography in dairy cows. J. Dairy Sci., 95(2): 735-742. DOI: 10.3168/jds.2011-4762
Alsaaod, M., C. Syring, J. Dietrich, M.G. Doherr, T. Gujan and A. Steiner. 2014. A field trial of infrared thermography as a non-invasive diagnostic tool for early detection of digital dermatitis in dairy cows. Vet. J., 199(2): 281-285. DOI: 10.1016/j.tvjl.2013.11.028
Bagate, M.S., J.K. Mahla, P.V. Parikh, D.B. Patil and M.U.D. Dar. 2012. Incidence of foot disorders in dairy animals - A retrospective study. Intas Polivet, 13(2): 192-194.
Damasceno, F.A., J.M. Viana, I.D.F.F. Tinoco, R.C.C. Gomes and L. Schiassi. 2010. Adaptação de bubalinos ao ambiente tropical. Revista Eletrônica Nutritime, 7(5): 1370-1381.
De Ruediger, F.R., P.H. Yamada, L.G.B. Barbosa, M.G.M. Chacur, J.C.P. Ferreira, N.A.T. de Carvalho, G.A.M. Soriano, V.M. Codognoto and E. Oba. 2018. Effect of estrous cycle phase on vulvar, orbital area and muzzle surface temperatures as determined using digital infrared thermography in buffalo. Anim. Reprod. Sci., 197: 154-161. DOI: 10.1016/j.anireprosci.2018.08.023.
Green, L.E., V.J. Hedges, Y.H. Schukken, R.W. Blowey and A.J. Packington. 2002. The impact of clinical lameness on the milk yield of dairy cows. J. Dairy Sci., 85(9): 2250-2256. DOI: 10.3168/jds.S0022-0302(02)74304-X
Hellebrand, H.J., U. Brehme, H. Beuche, U. Stollberg and H. Jacobs. 2003. Application of thermal imaging for cattle management. In Proceeding 1st European Conference on Precision Livestock Farming, Berlin, Germany. p. 761-763. Available on: http://www2.atb-potsdam.de/hauptseite-deutsch/Institut/Abteilungen/abt2/mitarbeiter/jhellebrand/jhellebrand/Publikat/ECPLF.pdf
Hess, E. 1904. Klauenkrankheitrn. In Bayer, J. and E. Frohner. Handbuch der Tierarztlichen chirurgie and Geburtshilfe. Wilhelm Braumuller, Wien. Available on: https://www.bing.com/accessed
Joshi, V.K. 2006. Prevalence of hoof diseases and their surgical management in cattle and buffaloes. M.V. Sc Thesis, Anand Agricultural University, Anand, India. p. 50-62.
Kameya, T. and S. Yamaoka. 1968. Effect of atmospheric conditions on skin temperature in horses. Exp. Rep. Equine. Heal., 5: 1-12. DOI: 10.11535/jes1961.1968.1
Koga, A., M. Sugiyama, A.N.D. Barrio, R.M. Lapitan, B.R. Arenda, A.Y. Robles and L. Cruz. 2004. Comparison of the thermoregulatory response of buffaloes and tropical cattle, using fluctuations in rectal temperature, skin temperature and haematocrit as an index. J. Agr. Sci., 142(3): 351-355. DOI: 10.1017/S0021859604004216
Landes, M., J. Cessna, L. Kuberka and K. Jones. 2017. India’s dairy sector: Structure, performance and prospects. Livestock, Dairy, and Poultry Outlook, Economic Research Service, U.S. Department of Agriculture, Washington DC., USA.
Lunstra, D.D. and G.H. Coulter. 1997. Relationship between scrotal infrared temperature patterns and natural-mating fertility in beef bulls. J. Anim. Sci., 75(3): 767-774.
Lutz, J., B. Molla, F. Silveira, W. Gebreyes and E.A. Lutz. 2011. Using infrared thermal imaging for mass screening of production animals for early detection of febrile diseases. Epidémiologie et Santé Animale, 59(60): 169-170.
Mogg, K.C. and C.C. Pollitt. 1992. Hoof and distal limb surface temperature in the normal pony under constant and changing ambient temperature. Equine Vet. J., 24(2): 134-139. DOI: 10.1111/j.2042-3306.1992.tb02798.x
Mohsina, A., M.M.S. Zama, P. Tamilmahan, M.B. Gugjo, K. Singh, A. Gopinathan, M. Gopi and K. Karthik. 2014. A retrospective study on incidence of lameness in domestic animals. Vet. World, 7(8): 601-604. DOI: 10.14202/vetworld.2014.601-604
Montanholi, Y.R., E. Odongo, K.C. Swanson and F.S. Schenkel. 2008. Application of infrared thermography as an indicator of heat and methane production and its use in the study of skin temperature in response to physiological events in dairy cattle (Bos taurus). J. Therm. Biol., 33(8): 468-475. DOI: 10.1016/j.jtherbio.2008.09.001
Palmer, S.E. 1983. Effect of ambient temperature upon the surface temperature of the equine limb. Am. J. Vet. Res., 44(6): 1098-1101.
Poikalainen, V., J. Praks, I. Veermae and E. Kokin. 2012. Infrared temperature patterns of cow’s body as an indicator for health control at precision cattle farming. Agronomy Research Biosystem Engineering, (Sppl. 1): 187-194. Available on: https://agronomy.emu.ee/vol10Spec1/p10s121.pdf
Polat, B., A. Colak, M. Cengiz, E. Yanmaz, H. Oral, A. Bastan, S. Kaya and A. Hayirli 2010. Sensitivity and specificity of infrared thermography in detection of subclinical mastitis in dairy cows. J. Dairy Sci., 93(8): 3525-3532. DOI: 10.3168/jds.2009-2807
Purwanto, B.P., Y. Abo, R. Sakamoto, F. Furumoto and S. Yamamoto. 1990. Diurnal patterns of heat production and heart rate under thermoneutral conditions in Holstein Friesian cows differing in milk production. J. Agr. Sci., 114(2): 139-142. DOI: 10.1017/S0021859600072117
Sathiyabarathi, M., S. Jeyakumar, A. Manimaran, H.A. Pushpadass, M. Sivaram, K.P. Ramesha and D.N. Das. 2016a. Thermographic imaging: A potential non-invasive technique for early detection of subclinical mastitis in crossbred dairy cows. In The 44th Dairy Industry Conference, Indian Council of Agricultural Research-National Dairy Research Institute, Karnal, India.
Sathiyabarathi, M., S. Jeyakumar, A. Manimaran, G. Jayaprakash, H.A. Pushpadass, M. Sivaram, K.P. Ramesha, D.N. Das, M.A. Kataktalware, M.A. Prakash and R.D. Kumar. 2016b. Infrared thermography: A potential noninvasive tool to monitor udder health status in dairy cows. Vet. World, 9(10): 1075-1081. DOI: 10.14202/vetworld.2016.1075-1081
Sathiyabarathi, M., S. Jeyakumar, A. Manimaran, H.A. Pushpadass, M. Sivaram, K.P. Ramesha, D.N. Das, M.A. Kataktalware, G. Jayaprakash and T.K. Patbandha. 2016c. Investigation of body and udder skin surface temperature differentials as an early indicator of mastitis in Holstein Friesian crossbred cows using digital infrared thermography technique. Vet. World, 9(12): 1386-1391. DOI: 10.14202/vetworld.2016.1386-1391
Sathiyabarathi, M., S. Jeyakumar, A. Manimaran, H.A. Pushpadass and M. Sivaram. 2017. Influence of machine milking on udder and teat surface temperature in Holstein Friesian crossbred cows monitored by digital infrared thermography, p. 313-316. In Proceedings of the 9th Kerala Veterinary Science Congress, India.
Sathiyabarathi, M., S. Jeyakumar, A. Manimaran, H.A. Pushpadass, M. Sivaram, K.P. Ramesha, D.N. Das and M.A. Kataktalware. 2018. Infrared thermal imaging of udder skin surface temperature variations to monitor udder health status in Bos indicus (Deoni) cows. Infrared Physics and Technology, 88: 239-244. DOI: 10.1016/j.infrared.2017.11.028
Schaefer, A.L., S.D.M. Jones, A.K.W. Tong and B.C. Vincent. 1998. The effects of fasting and transportation on beef cattle. 1. acid-base-electrolyte balance and infrared heat loss of beef cattle. Livest. Prod. Sci., 20: 15-24. DOI: 10.1016/0301-6226(88)90050-4
Schaefer, A.L., N. Cook, S.V. Tessaro, D. Deregt, G. Desroches, P.L. Dubeski, A.K.W. Tong and D.L. Ggodson. 2004. Early detection and prediction of infection using infrared thermography. Can. J. Anim. Sci., 84(1): 73-80. DOI: 10.4141/A02-104
Schaefer, A.L., N.J. Cook, J.S. Church, J. Basarab, B. Perry, C. Millar and A.K.W. Tong. 2007. The use of infrared thermography as an early indicator of bovine respiratory disease complex in calves. Res. Vet. Sci., 83(3): 376-384. DOI: 10.1016/j.rvsc.2007.01.008
Smilie, R.H., K.H. Hoblet, W.P. Weiss, M.L. Estridge, D.M. Rings and G.L. Sachnithkey. 1996. Prevalence of lesions associated with subclinical laminitis in first-lactation cows from herds with high milk production. J. Am. Vet. Med. Assoc., 208(9): 1445-1451.
Spiers, D.E., J.N. Spain, J.D. Sampson and R.P. Rhoads. 2004. Use of physiological parameters to predict milk yield and feed intake in heat-stressed dairy cows. J. Therm. Biol., 29: 759-764. DOI: 10.1016/j.jtherbio.2004.08.051
Stewart, M., J.R. Webster, A.L. Schaefer, N.J. Cook and S.L. Scott. 2005. Infrared thermography as a non-invasive tool to study animal welfare. Anim. Welfare, 14(4): 319-325.
Stewart, M., G.A. Verkerk, K.J. Stafford, A.L. Schaefer and J.R. Webster. 2010. Noninvasive assessment of autonomic activity for evaluation of pain in calves, using surgical castration as a model. J. Dairy Sci., 93(8): 3602-3609. DOI: 10.3168/jds.2010-3114
Turner, T.A. 2001. Diagnostic thermography. Vet. Clin. N. Am. Equine, 17(1): 95-113. Doi: 10.1016/S0749-0739(17)30077-9
Whay, H.R. and D.C.J. Main. 2003. Assessment of the welfare of dairy cattle using animal-based measurements: direct observations and investigation of farm records. Vet. Rec., 153(7): 197-202. DOI: 10.1136/vr.153.7.197








.png)








