Expression and structural variations of toll-like receptor 4 gene in buffalo and cattle
DOI:
https://doi.org/10.56825/bufbu.2022.4113823Keywords:
Bubalus bubalis, buffaloes, innate immunity, TLR4, LPS, cattle, geneAbstract
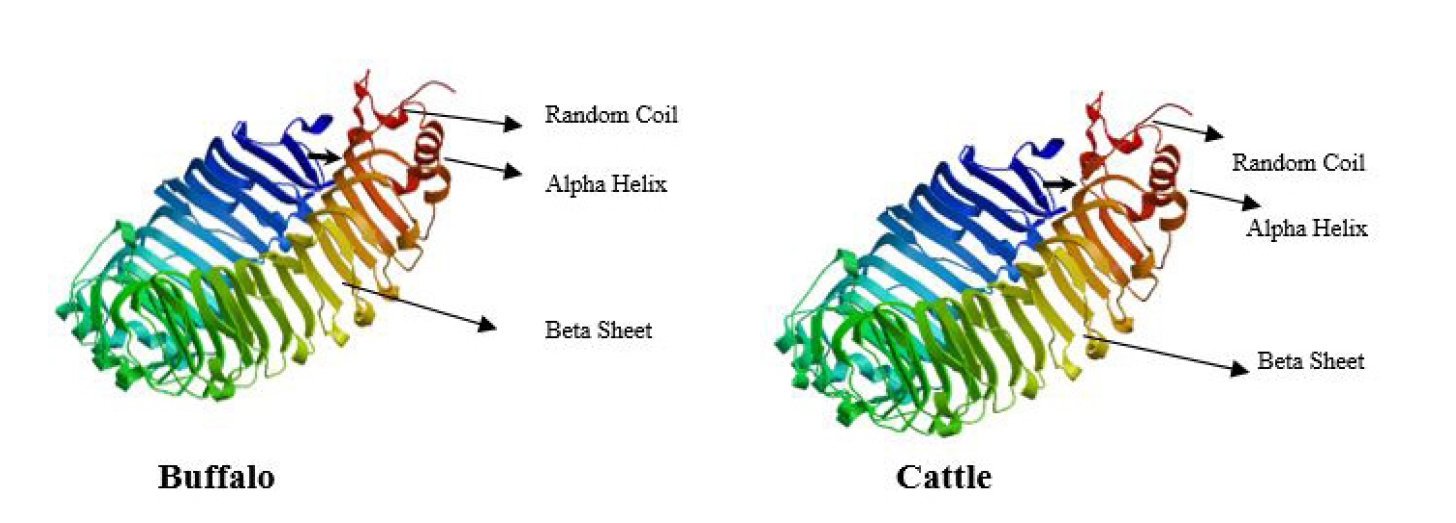
Systematic studies on expression and characterization of toll like receptors (TLRs) in buffalo and cattle will help to understand the innate immune response specific to each species of animals. TLR4 gene is one of the most studied innate immune receptors which is mainly triggered by lipopolysaccharide. Therefore, the present study was carried out to investigate the mRNA expression and sequence analysis of TLR4 in buffalo in comparison to that of cattle. In vitro expression of TLR4 was assessed by challenging the Peripheral Blood Mononuclear Cells (PBMC) with bacterial LPS which showed significant increase of TLR4 gene expression (P≤0.01) in buffaloes when compared to cattle. Further the comparison of mRNA sequence of TLR4 of buffalo and cattle revealed 96% similarity with the cattle sequence. The primary structure of protein showed the highest percent of leucine amino acid for both the species of animals. At the same time, the number of Leucine Rich repeats (LRR) of buffalo is varied from those present in cattle TLR4. Alpha helix is the prominent secondary structure, however, there were few amino acid variations between buffalo and cattle in certain regions of TLR4 which altered the three dimensional conformation of TLR4 proteins, this in-turn possibly can influence the binding affinity and interaction with pathogens which may influence the difference in immune response in buffalo when compared to that of cattle.
Downloads
Metrics
References
Benakanakere, M.R., Q. Li, M.A. Eskan, A.V. Singh, J. Zhao, J.C. Galicia, P. Stathopoulou, T.B. Knudsen and D.F. Kinane. 2009. Modulation of TLR2 protein expression by miR-105 in human oral keratinocytes. J. Biol. Chem., 284(34): 23107-23115. DOI: 10.1074/jbc.M109.013862.
Bi, D., Y. Wang, Y. Gao, X. Li, Q. Chu, J. Cui and T. Xu. 2018. Recognition of lipopolysaccharide and activation of NF-κB by cytosolic sensor NOD1 in Teleost fish. Front. Immunol., 9: 1413. DOI: 10.3389/fimmu.2018.01413.
Borrielo, G., R. Capparelli, M. Bianco, D. Fenizia, F. Alfano, F. Capuano, D. Ercolini, A. Parisi and S. Roperto. 2006. Genetic resistance to Brucella abortus in the Water buffalo (Bubalus bubalis). Infect. Immun., 74(4): 2115-2120. DOI: 10.1128/IAI.74.4.2115–2120.2006
Dos Santos, L.S., J.C. Sa, D.L. Dos Santos Ribeiro, N.P. Chaves, J.P. da Silva Mol, R.L. Santos, T.A. da Paixao and A.V.de Carvalho Neta. 2017. Detection of Brucella sp. infection through serological, microbiological, and molecular methods applied to buffaloes in Maranhao state, Brazil. Trop. Anim. Health Pro., 49(4): 675-679. DOI: 10.1007/s11250-017-1238-3
Dubey, P.K., S. Goyal, P. Kathiravan, B.P Mishra, S.K. Gahlawat and R.S. Kataria. 2013. Sequence characterization of River buffalo Toll-like receptor genes 1-10 reveals distinct relationship with cattle and sheep. Int. J. Immunogenet., 40(2): 140-148. DOI: 10.1111/j.1744- 313X.2012.01135.x
Griesbeck-Zilch, B., H.H.D. Meyer, C. Kühn, M. Schwerin and O. Wellnitz. 2008. Staphylococcus aureus and Escherichia coli cause deviating expression profiles of cytokines and lactoferrin messenger ribonucleic acid in mammary epithelial cells. J. Dairy. Sci., 91(6): 2215-2224. DOI: 10.3168/jds.2007-0752.
Hoshino, K., O. Takeuchi, T. Kawai, H. Sanjo, T. Ogawa, Y. Takeda, K. Takeda and S. Akira. 1999. Cutting edge: Toll-like receptor 4 (TLR 4)-deficient mice are hyporesponsive to lipopolysaccharide: evidence for TLR 4 as the LPS gene product. J. Immunol., 162(7): 3749-3752. Available on: http://www.jimmunol.org/content/162/7/3749
Ibeagha-Awemu, E.M., J.W. Lee, A.E. Ibeagha, D.D. Bannerman, M.J. Paape and X. Zhao. 2008. Bacterial lipopolysaccharide induces increased expression of Toll-like receptor (TLR) 4 and downstream TLR signaling molecules in bovine mammary epithelial cells. Vet. Res., 39(2): 11. DOI: 10.1051/vetres:2007047
Iqbal, M. and V.J. Philbin. 2005. Expression patterns of chicken Toll-like receptor mRNA in tissues, immune cell subset and cell lines. Vet. Immunol. Immunopathol., 104(1-2): 117-127. DOI: 10.1016/j.vetimm.2004.11.003.
Lamping, N., R. Dettmer, N.W. Schroder, D. Pfeil, W. Hallatschek, R. Burger and R.R. Schumann. 1998. LPS-binding protein protects mice from septic shock caused by LPS or gram-negative bacteria. J. Clin. Invest., 101(10): 2065-2071. DOI: 10.1172/JCI2338
Livak, K.J. and T.W. Schmittgen. 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods, 25(4): 402-408. DOI: 10.1006/meth.2001.1262
Menzies, M. and A. Ingham. 2006. Identification and expression of Toll-like receptors 1-10 in selected bovine and ovine tissues. Vet. Immunol. Immunop., 109(1-2): 23-30. DOI: 10.1016/j.vetimm.2005.06.014.
Nardi, G.J., M.G. Ribeiro, A.M. Jorge, J. Megid and L.M.P. Silva. 2012. Serological profile of buffalo (Bubalus bubalis) female calves vaccinated with standard Brucella abortus strain 19 using rose Bengal, 2-mercaptoethanol and complement fixation test. Biologicals, 40(2): 158-161. DOI: 10.1016/j.biologicals.2011.09.016.
Nisha, P., J. Thanislass, P.X. Antony, H.K. Mukhopadhyay and K.V.S. Reddy. 2013. Bacterial DNA induced TNF-α expression in buffaloes (Murrah) in comparison to that of cross breed cattle. Journal of Buffalo Science, 2(1): 8-11. DOI: 10.6000/1927-520X.2013.02.01.2
Panigrahi, M., A. Sharma and B. Bhushan. 2014. Molecular characterization and expression profile of partial TLR4 gene in association to mastitis in crossbred cattle. Anim. Biotechnol., 25(3): 188-199. DOI: 10.1080/10495398.2013.856797
Petzl, W., H. Zerbe, J. Günther, W. Yang, H.M. Seyfert, G. Nürnberg and H.J. Schuberth. 2008. Escherichia coli, but not Staphylococcus aureus triggers an early increased expression of factors contributing to the innate immune defense in the udder of the cow. Vet. Res., 39(2): 18. DOI: 10.1051/vetres:2007057.
Poltorak, A., X. He, I. Smirnova, M.Y. Liu, H.C. Van, X. Du, D. Birdwell, E. Alejos, M. Silva, C. Galanos, M. Freudenberg, P. Ricciardi-Casatagnoli, B. Layton and B. Beutler. 1998. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: Mutations in Tlr4 gene. Science, 282(5396): 2085-2088. DOI: 10.1126/science.282.5396.2085
Prueet, S.B., Q. Zheng, R. Fan, K. Matthews and C. Schwab. 2004. Acute exposure to ethanol affects Toll-like receptor signaling and subsequent responses: An overview of recent studies. Alcohol, 33(3): 235-239. DOI: 10.1016/j.alcohol.2004.08.003
Rabindra, N.B. and S. Akira. 2005. Toll-like receptor signaling: Emerging opportunities in human diseases and medicine. Current Immunology Reviews, 1(1): 81-90. DOI: 10.2174/ 1573395052952897
Takeda, K. and S. Akira. 2005. Toll-like receptors in innate immunity. Int. Immunol., 17(1): 1-14. DOI: 10.1093/intimm/dxh186.
Thanislass, J., G. Yuvaraj and K.V. Subba Reddy. 2009. Characterization of TLR4 signaling in Water buffalo (Bubalus Bubalis). Vet. Res. Commun., 33(2): 97-102. DOI: 10.1007/s11259-008-9074-6
Vignesha, A.R., S. Dhanasekarana, G.D. Raja, C. Balachandranb, N. Pazhanivelb, C. Sreekumarc, K.G. Tirumurugaana, A. Rajaa and K. Kumanana. 2012. Transcript profiling of pattern recognition receptors in a semi domesticated breed of buffalo, Toda, of India. Vet. Immunol. Immunop., 147(1-2): 51-59. DOI: 10.1016/j.vetimm. 2012.02.009
Zarember, K.A and P.J. Godowski. 2002. Tissue expression of human toll-like receptors and differential regulation of toll-like receptor mRNAs in leukocytes in response to microbes, their products, and cytokines. J. Immunol., 168(2): 554-561. DOI: 10.4049/ J Immunol.168.2.554.








.png)








