Isolation and molecular detection of biofilm producing and multiple drug resistant Enterococcus faecalis from the buffalo meat
DOI:
https://doi.org/10.56825/bufbu.2022.4134917Keywords:
Bubalus bubalis, buffaloes, antimicrobial resistance, biofilm, buffalo meat, carabeef, ddlE gene, Enterococcus faecalis, PCR, vancomycin resistant enterococciAbstract
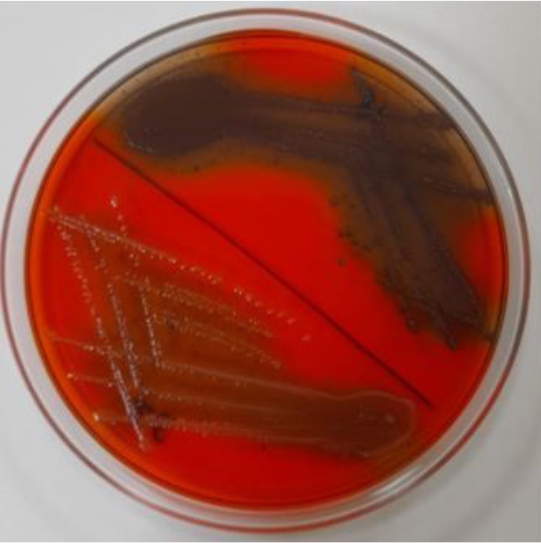
The present study was conducted to determine the drug resistance pattern and the occurrence of biofilm producing Enterococcus faecalis from buffalo meat samples collected from in and around the Anand city, Gujarat. A total of 100 samples (meat, hand’s swab, knife’s swab) were collected aseptically from the butcher shops. Out of 100 samples, 52 (52%) samples were found to be positive on selective media, which were subjected to polymerase chain reaction revealing that 40 isolates were of Enterococcus faecalis. Antibiotic sensitivity test showed that all the isolates were sensitive to Ampicillin (100%); sensitive to intermediate for Gentamicin and resistance was observed against Tigecycline (85%), Trimethoprim (60%), Vancomycin (50%), Norfloxacin (37.5%), and Imipenem-cilastatin (25%). Out of all the PCR positive isolates, 95% (38/40) were biofilm producers when observed phenotypically on Congo Red Agar (CRA). So, it can be concluded that the buffalo meat can be a possible intermediary vehicle for the spread of multidrug-resistant biofilm producing enterococci strains to humans.
Downloads
Metrics
References
Costerton, J.W. and P.S. Stewart. 2001. Battling biofilms. Sci. Am., 285(1): 74-81. DOI: 10.1038/scientificamerican0701-74
Dutka-Malen, S., S. Evers and P. Courvalin. 1995. Detection of glycopeptide resistance genotypes and identification to the species level of clinically relevant enterococci by PCR. J. Clin. Microbiol., 33(1): 24-27. DOI: 10.1128/JCM.33.1.24-27.1995
Esmaeili, Z., J. Sadeghi, S. Razavi, M. Oshaghi, S. Sayyahfar, M. Rahbar and M. Talebi. 2018. High level of biofilm formation and virulence factors in enterococci species isolated from clinical and normal flora samples. Infect. Dis. Clin. Prac., 26(1): 27-30. DOI: 10.1097/IPC.0000000000000519
Fallah, F., M. Yousefi, M.R. Pourmand, A. Hashemi, A.N. Alam and D. Afshar. 2017. Phenotypic and genotypic study of biofilm formation in Enterococci isolated from urinary tract infections. Microb. Pathogenesis, 108: 85-90. DOI: 10.1016/j.micpath.2017.05.014
Fischetti, V., R. Novick, J. Ferretti, D. Portnoy, J. Rood and M. Park. 2006. Gram-Positive Pathogens, 2nd ed. ASM Press, Washington DC., USA. 888p.
Fisher, K. and C. Phillips. 2009. The ecology, epidemiology and virulence of Enterococcus. Microbiology Microbiology Society, 155(6): 1749-1757. DOI: 10.1099/mic.0.026385-0
Gomes, B.C., C.T. Esteves, I.C.V. Palazzo, A.L.C. Darini, G.E. Felis, L.A. Sechi, B.D.G.M. Franco and De E.C.P. De Martinis. 2008. Prevalence and characterization of Enterococcus spp. isolated from Brazilian foods. Food Microbiol., 25(5): 668-675. DOI: 10.1016/j.fm.2008.03.008
Hayes, J.R., L.L. English, P.J. Carter, T. Proescholdt, K.Y. Lee, D.D. Wagner and D.G. White. 2003. Prevalence and antimicrobial resistance of enterococcus species isolated from retail meats. Appl. Environ. Microb., 69(12): 7153-7160. DOI: 10.1128/AEM.69.12.7153-7160.2003
Kasimoglu-Dogru, A., Y.E. Gencay and N.D. Ayaz. 2010. Prevalence and antibiotic resistance profiles of Enterococcus species in chicken at slaughter level; Absence of van A and van B genes in E. faecalis and E. faecium. Res. Vet. Sci., 89(2): 153-158. DOI: 10.1016/j.rvsc.2010.02.005
Kročko, M., M. Čanigová, V. Ducková, A. Artimová, J. Bezeková and J. Poston. 2011. Antibiotic resistance of Enterococcus species isolated from raw foods of animal origin in South West part of Slovakia. Czech J. Food Sci., 29(6): 654-659. DOI: 10.17221/246/2010-cjfs
Lewis, K. 2001. Riddle of biofilm resistance. Antimicrob. Agents Ch., 45(4): 999-1007. DOI: 10.1128/AAC.45.4.999-1007.2001
Maccallum, W.G. and T.W. Hastings. 1899. A case of acute endocarditis caused by micrococcus zymogenes (nov. spec.), with a description of the microorganism. J. Exp. Med., 4(5-6): 521-534. DOI: 10.1084/jem.4.5-6.521
Majhenič, A.Č., I. Rogelj and B. Perko. 2005. Enterococci from tolminc cheese: Population structure, antibiotic susceptibility and incidence of virulence determinants. Int. J. Food Microbiol., 102(2): 239-244. DOI: 10.1016/j.ijfoodmicro.2004.12.021
Manero, A. and A.R. Blanch. 1999. Identification of Enterococcus spp. with a biochemical key. Appl. Environ. Microb., 65(10): 4425-4430. DOI: 10.1128/aem.65.10.4425-4430.1999
Olsen, R.H., H.C. Schønheyder, H. Christensen and M. Bisgaard. 2012. Enterococcus faecalis of human and poultry origin share virulence genes supporting the zoonotic potential of E. faecalis. Zoonoses Public Hlth., 59(4): 256-263. DOI: 10.1111/j.1863-2378.2011.01442.x
O’Neill, J. 2016. Tackling drug-resistant infections globally: Final report and recommendations. The Review on Antimicrobial Resistance, Government of the United Kingdom, UK. Available on: https://apo.org.au/sites/default/files/resource-files/2016-05/apo-nid63983.pdf
Sharvari, S.A. and P.G. Chitra. 2012. Evaluation of different detection methods of biofilm formation in clinical isolates of Staphylococci. International Journal of Pharma and Bio Sciences, 3(4): 724-733. Available on: https://www.scielo.br/pdf/bjid/v15n4/v15n4a02.pdf
Sundsfjord, A., G.S. Simonsen and P. Courvalin. 2001. Human infections caused by glycopeptide-resistant Enterococcus spp: Are they a zoonosis? Clin. Microbiol. Infec., 7(4): 16-33. DOI: 10.1046/j.1469-0691.2001.00055.x
Wheeler, A.L., P.G. Hartel, D.G. Godfrey, J.L. Hill and W.I. Segars. 2002. Potential of Enterococcus faecalis as a human fecal indicator for microbial source tracking. J. Environ. Qual., 31(4): 1286-1293. DOI: 10.2134/jeq2002.1286








.png)








