Incidence of hydatid disease in buffaloes
Keywords:
buffaloes, Hydatidosis, Bubalus bubalis, Incidence, hydatidosis, Buffaloes, incidence, PCRAbstract
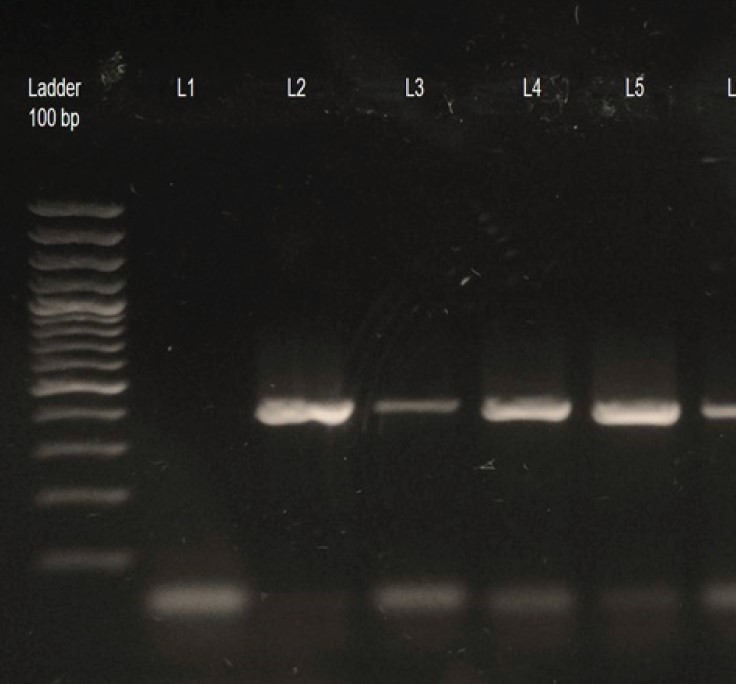
The present study on incidence of hydatid disease in buffaloes was conducted to evaluate the incidence of hydatid cyst grossly as well as by means of polymerase chain reaction in buffaloes. Results indicated an overall incidence of 12% of hydatid disease in buffaloes in Akola district of Maharashtra. Liver (75%) showed the most predominant site as compared to lung. All most all the cysts were of small size (87.50%) having below 5 cm in diameter and are single in organ. Most of the hydatid cysts in liver (62.50%) did not revealed protoscolices hence considered as sterile. Each PCR isolate obtained from individual hydatdi cyst were further showed 434 base pair fragment of the mitochondrial cytochrome oxidase -1 gene on 1.5% agarose gel electrophoresis and confirmed the hydatid disease.
Downloads
Metrics
References
Barnes, T. S., L. A. Hinds, D .J. Jenkins and G. T. Coleman. 2007. Precocious development of hydatid cysts in a macropodid host. Int. J. Parasitol. 37: 1379-1389.
-----------------------------------------------------------------------
Benner, C., H. Carabin, L. P. S. Serrano, C. M. Budkeand and D. Carmena. 2010. Analysis of the economic impact of cystic echinococcosis in Spain. Bull World Hlth Or. 88: 49 - 57.
----------------------------------------------------------------------
Bhattacharya, D., A. K. Bera, B. C. Bera, A. Maity and S. K. Das. 2007. Genotypic characterization of Indian cattle, buffalo and sheep isolates of Echinococcus granulosus. Vet Parasitol. 143: 371–374.
---------------------------------------------------------------------
Dinkel, A., E. M. Njoroge, A. Zimmermann, M. Walz, E. Zeyhle, I. E. Elmahdi, U. Mackenstedt, T. Romig. 2004. A PCR system for detection of species and genotypes of the Echinococcus granulosus-complex, with reference to the epidemiological situation in eastern Africa. Int. J. Parasitol. 34 : 645 – 653.
------------------------------------------------------------------------
Dhote, S. W., Sadhana Patil, R. D. Sadekar, M. V. Joshi and S. S. Bhagwat. 1992. Incidence of morbid conditions in livers of slaughtered bullocks. Indian Journal of Animal Sciences. 62 (8): 744-746.
-----------------------------------------------------------------------
Getaw, A., D. Beyene, D. Ayana, B. Megersa and F. Abunna. 2010. Hydatidosis: Prevalence and its economic importance in ruminants slaughtered at Adama municipal abattoir, Central Oromia, Ethiopia. Acta Tropica, 113 : 221 – 225.
----------------------------------------------------------------------
Kebede, N., A. Mitiku and G. Tilahun. 2009. Hydatidosis of slaughtered animals in Bahir Dar Abattoir, Northwestern Ethiopia. Trop Anim Health Prod. 41 : 43-50.
----------------------------------------------------------------------
Kurkure, N. V., S. W. Dhote, A. G. Bhandarkar,M. V.Joshi and D. L. Paikne. 1992. A note on incidence of hydatidosis in cattle. PKV Res. J. 16 : 95.
----------------------------------------------------------------------
McManus, D. P. 2006. Molecular determination of taeniid cestodes. Parasitol. Int. 55 : 31 – 37.
----------------------------------------------------------------------
Melaku, A., B. Lukas and B. Bogale. 2012. Cyst viability organ distribution and financial losses due to hydatidosis in cattle slaughtered at Dessie Munciple Abattoir, North-eastern Ethopia. Vet. World. 5 : 213-218.
----------------------------------------------------------------------
Moro, P. L., M. Nakao, M. Ito, P. M. Schantz, C. Cavero and L. Cabrera. 2009. Molecular identification of Echinococcus isolates from Peru. Parasitol. Int. 58 : 184 – 186.
-----------------------------------------------------------------------
Pathak, V. P., D. R. Rajgude, M. V. Joshi, U. B. Deore and R. S. Ingole. 2004. Incidence of hydatid cysts in domastic runinants- a post-mortem study. Indian Vet. Med. Jour. 28 : 173-175.
----------------------------------------------------------------------
Pedenekar, R. P., M. L. Gatne, R. C. Thompson, R. J. Traub. 2009. Molecular and morphological characterization of Echinococcus from food producing animals in India. Journal of Vet. Parasiolo. 165 : 58-65.
---------------------------------------------------------------------
Sanchez, E., O. Caceres, C. Naquira, D. Garcia, G. Patino, H. Silvia, A. C. Colotao, and O. Fernanes. 2010. Molecular characterization of Echinococcus granulosus from Peru by sequencing of the mitochondrial cytochrome C oxidase subunit 1 gene. Mem Inst Oswaldo Cruz, Rio Janeiro. 105 : 806 – 810.
-----------------------------------------------------------------------
Sharma, M., R. Sehgal, B. A. Fomda, A. Malhotra, and N. Malla. 2013. Molecular characterization of Echinococcus granulosus cysts in North Indian patients: Identification of G1, G3, G5 and G6 Genotypes. PLOS, Neglected Tropical Diseases. 7 : 1 - 6.
----------------------------------------------------------------------
Sheeba, A., A. Sangaran, B. R. Latha and A. Raja. 2016. Incidence and fertility status of hydatid cysts in buffaloes. Buffalo Bulletin. 35 : 379-381.
---------------------------------------------------------------------
Singh, B. B., N. K. Dhand, S. Ghatak and J. P. Gill. 2014. Economic losses due to cystic echinococcus in India: Need for urgent action to control the disease. Prev Vet Med. 113 : 1-12.
---------------------------------------------------------------------
Vural, S., H. Kales and M. Haligur. 2004. Unilocular spleenic hydatidosis in a Sheep. The Internet Journal of Veterinary Medicine. 2: 1 – 6.




.png)








